Introduction to Prions
Structure |
What it does |
|
The prion is unlike any other pathogen, in that it does not need nucleic acids to survive. Therefore, it is not living, it is an even simpler structure than a virus.
|
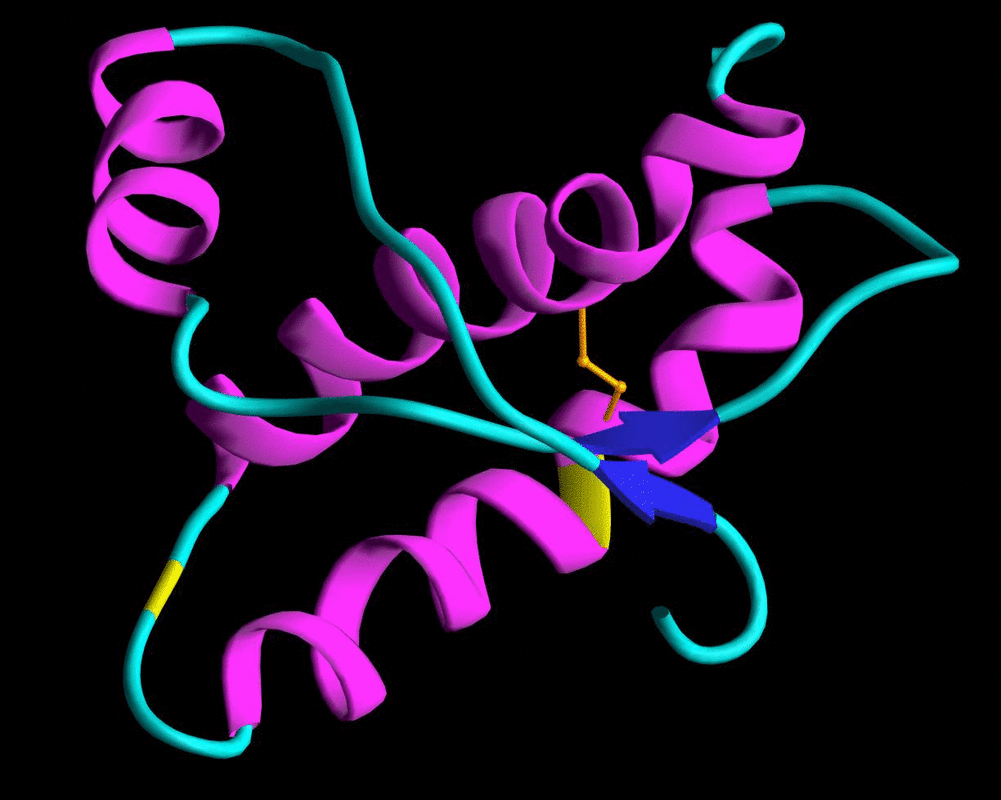
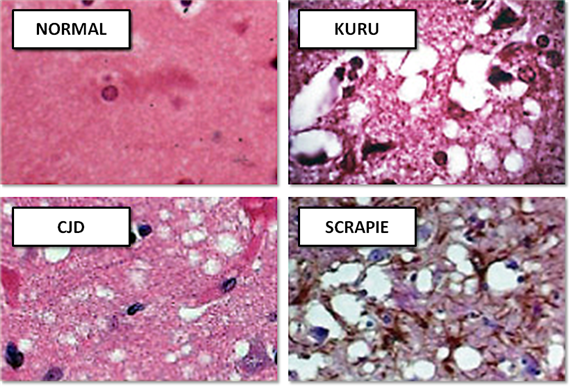
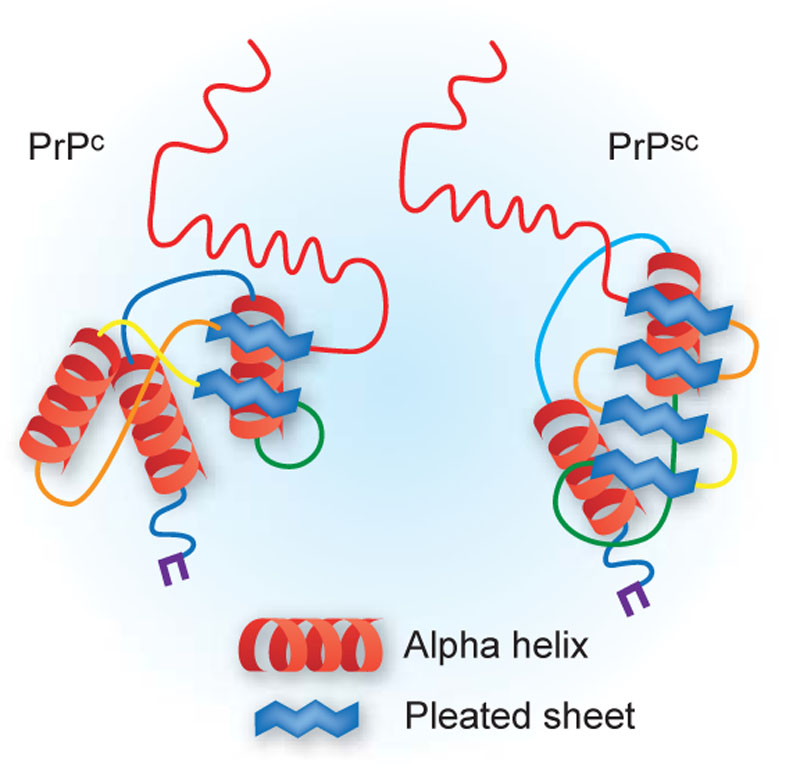
The prion in its normal form (PrP) is a harmless protein that is found in the brains of many vertebrates. However, when a misfolded protein changes its structure, it causes tiny holes to form in the brain.
|
Prions are microscopic pathogens that were discovered in the late 20th century. This class of diseases, caused by the infectious protein PrPSc, cause TSEs, or Transmissible Spongiform Encephalopathies. Because prions do not rely on DNA or RNA to replicate or function, they cannot be killed with treatments intended for other pathogens. It is considered by some to be an indestructible pathogen. There is some fact behind this notion, as cooking meat from an infected cow will not make it safe. It can not be killed by normal sterilization in hospitals, as it will survive formaldehyde, the intense temperatures of an autoclave, and will slip in between the UV rays from sterilizing air filters. Scrapie prions have been shown to remain infective after 30 years in the soil of pastures where infected sheep or goats once roamed. The size of the prion makes it elusive, and some of the first researchers looking for the pathogen realized it was far too small to be a virus or bacteria. The hardiness of the prion and its size make it extremely hard to kill.
Misfolded Proteins
Misfolded proteins are the killers behind of some of the most costly neurodegenerative diseases such as Alzheimer's, Parkinson's, Huntington's, and ALS. Although Alzheimer's has the possibility of spreading from person to person through brain surgeries, prions are the only truly infectious diseases of the bunch. They are able to spread through eating infected tissues, hormonal supplements, certain organ transplants, and possibly blood. Chronic Wasting Disease can even spread through saliva.
References:
Figure 1 - Representation of the Cellular (PrPc) and Pathological (PrPSc) Isoforms of Prion Proteins. Digital image. Nature. N.p., 2009. Web. 12 July 2016. <http://www.nature.com/neuro/journal/v12/n1/fig_tab/nn0109-7_F1.html>.